Omar D. Abdul Sattar; Rozida Mohd Khalid; Siti Fairus M. Yusoff
Abstract
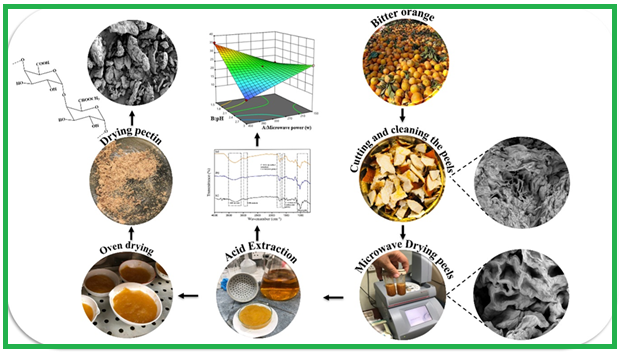
Pectin was extracted from bitter orange peels by subjecting them to electromagnetic field treatment and utilizing citric acid as an acidic catalyst. Fourier-Transform Infrared Spectroscopy ...
Read More
Pectin was extracted from bitter orange peels by subjecting them to electromagnetic field treatment and utilizing citric acid as an acidic catalyst. Fourier-Transform Infrared Spectroscopy (FTIR) analysis verified the presence of distinct functional groups in the pectin samples extracted using acidic catalysts, which exhibited spectra similar to commercially available pectin mentioned in previous studies. Scanning electron micrographs demonstrated that subjecting bitter orange peel to microwave heating led to the destruction of parenchymal cells. Furthermore, pectin samples were examined using scanning electron microscopy (SEM), revealing their structural changes. The extraction procedure was optimized using a multi-objective strategy based on yield-response surface techniques and the esterification degree. The experimental design employed a central composite design with five levels. The key variables under consideration were microwave power (ranging from 150 to 450 W) and solvent pH (ranging from 1.5 to 3.00). Optimal conditions were determined at a microwave power of 450 W and a solvent pH of 1.5, resulting in the highest yield (36.02%) and esterification degree (45.4) of pectin. The applied extraction model and its correlation were significant, as shown by the validation of the optimization model, which exhibited less than a 10% discrepancy between experimental and anticipated outcomes. To improve extraction efficiency, it is recommended to microwave fresh pectin raw materials before drying.